 
As with the alkanes, an increased amount of LDFs in alcohol containing molecules also causes in increase in boiling point. As we saw with the alkanes, the larger the carbon chain, the more LDFs that are present within the molecule. In addition to hydrogen bonding, alcohol molecules also have LDFs that occur between the nonpolar portions of the molecules. This is because molecules may have more than one type of intermolecular interactions. Only then can the molecule escape from the liquid into the gaseous state.Īnother interesting trend is apparent in table 9.1, is that as the alcohol molecules have more carbons, they also have higher boiling points. If the molecules interact through hydrogen bonding, a relatively large quantity of energy must be supplied to break those intermolecular attractions. The boiling point is a rough measure of the amount of energy necessary to separate a liquid molecule from its nearest neighbors. Since alcohols have the capacity to form hydrogen bonds, their boiling points are significantly higher when compared to hydrocarbons of comparable molar mass. In contrast, if we analyze the compounds that contain an alcohol functional group, even methanol (with only one carbon atom) is a liquid at room temperature. The boiling points of alkanes with one to four carbon atoms are so low that all of these molecules are gases at room temperature. Table 9.1 Comparison of Molar Mass and Boiling PointsĪlkanes are nonpolar and are thus associated only through relatively weak London Dispersion Forces (LDFs). The table shows that substances with similar molar masses can have quite different boiling points. 1 lists the molar masses and the boiling points of some common compounds. Recall that physical properties are determined to a large extent by the type of intermolecular forces. The OH groups of alcohol molecules make hydrogen bonding possible. 2).įigure 9.2 Intermolecular Hydrogen Bonding in Methanol. Replacing a hydrogen atom from an alkane with an OH group allows the molecules to associate through hydrogen bonding (Figure 9. 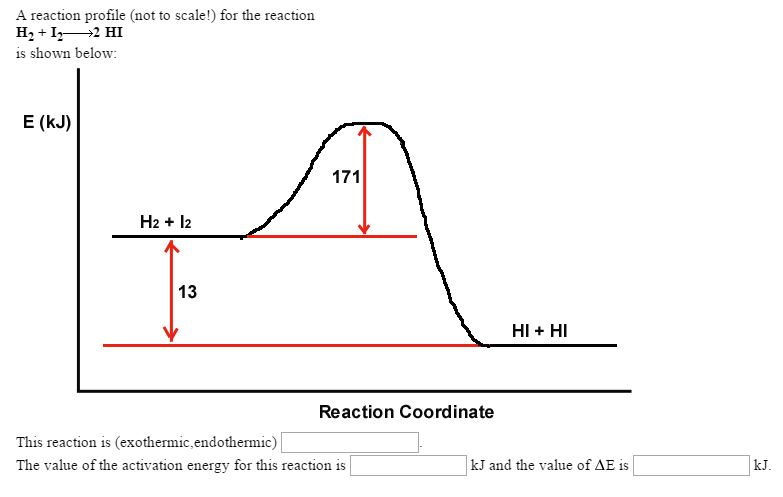  
This relationship is particularly apparent in small molecules and reflected in the physical and chemical properties of alcohols with low molar mass. Like the H–O–H bond in water, the R–O–H bond is bent, and the -OH portion of alcohol molecules are polar.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
